インフルエンザワクチンと免疫チェックポイント阻害剤
がん化学療法を受けている患者にインフルエンザ予防接種を勧めるのはがん診療の基本中の基本ですが、従来のがん治療は易感染性が問題となることが多かったのに対して、最近増えてきた免疫チェックポイント阻害剤による治療は自己免疫疾患の惹起が問題となることがあるため免疫賦活する予防接種との安全性については新たな心配が出てきているようです。
以下に、PubmedやTwitterで検索したICIとインフルエンザワクチンに関する報告をまとめました。(本当はワクチン接種のシーズンより前にまとめようと思っていたので遅くなってしまいました)
ICIでirAEは増加するのか
2018年には127人の肺癌患者での観察から、ICIの投与中の患者に対するインフルエンザワクチンを行ってもirAEは増加しないらしいとの報告が出ています。症例数が各群2桁と若干少なめであることから重症irAE発症のリスク比の95%信頼区間が非常に広い(ratio 2.07、95% CI 0.28-15.43)ことに注意が必要です。/Eur J Cancer
Influenza vaccination in patients with lung cancer receiving anti-PD-1 immunotherapy does not induce irAEs in our cohort. With this result, influenza vaccination should not be deterred from this group of patients.
https://pubmed.ncbi.nlm.nih.gov/30368069/
イタリアのINDIVIa試験も症例数がそれほど多いわけではない検討ですが、irAEはともかくインフルエンザ発症率が予防接種群であまり下がっていませんね。これはどう考えたらよいのか…。/Immunotherapy
Although influenza vaccine may be clinically ineffective in advanced cancer patients receiving CKI, it seems not to negatively impact the efficacy of anticancer therapy.
https://pubmed.ncbi.nlm.nih.gov/30326787/
2019年に、免疫チェックポイント阻害薬を受けている癌患者にもインフルエンザ不活化ワクチンは安全との報告が出されています。。/Clin infect dis
For cancer patients who received flu vaccination within 65 days of initiating anti-PD-1 agents, rate of therapy related immune events was comparable to publishe
https://academic.oup.com/cid/article-abstract/70/2/193/5381697
ICI投与中の患者でインフルエンザワクチンは有効なのか
ICIを使用中の患者に対してインフルエンザワクチンを摂取した場合に抗体が問題なく付くのかという点では、CIDに報告があります。seroprotectionおよびseroconversionの割合はICI群が通常化学療法群より高かったという結果であり、ICI投与中の患者でもインフルエンザワクチンの効果は十分保たれそうです。/Clin infect dis
Abstract. Among prospectively enrolled adult patients with cancer receiving immune checkpoint inhibitors (ICIs; n = 46) or cytotoxic agents (n = 90), seroprotec
https://academic.oup.com/cid/article-abstract/71/2/422/5611338
インフルエンザワクチンでICIの効果が高まるか(基礎実験)
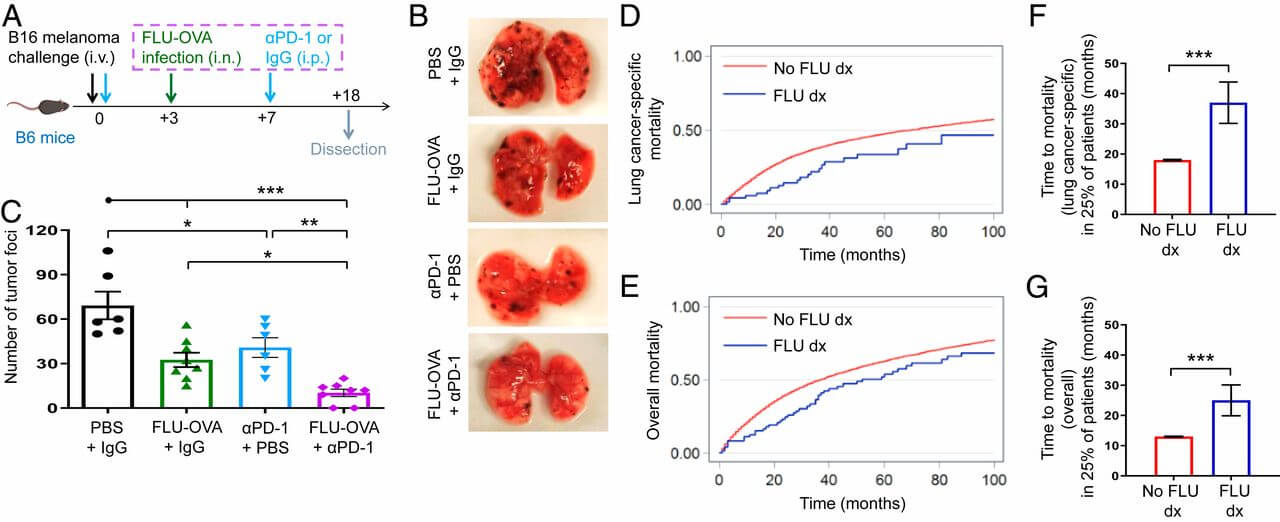
基礎実験では興味深いデータもあります。腫瘍病変内にインフルエンザワクチンを打つことでいわゆる免疫学的Cold tumorをHot tumorに変えることができるとのこと。一時期MET阻害剤でTILを増やす試みなどが考えられていましたが、免疫原性が高いワクチンを腫瘍内に打つことで炎症細胞浸潤をわざと引き起こすというのは面白い着眼点です。まだ基礎実験レベルの検討なので、今後の議論の発展があるのかどうか注視が必要です。/PNAS
Immunotherapy has revolutionized cancer treatment, yielding unprecedented long-term responses and survival. However, a significant proportion of patients remain refractory, which correlates with the absence of immune-infiltrated (“hot”) tumors. Here, we observed that FDA-approved unadjuvanted seasonal influenza vaccines administered via intratumoral injection not only provide protection against active influenza virus lung infection, but also reduce tumor growth by increasing antitumor CD8+ T cells and decreasing regulatory B cells within the tumor. Ultimately, intratumoral unadjuvanted seasonal influenza vaccine converts immunologically inactive “cold” tumors to “hot,” generates systemic responses, and sensitizes resistant tumors to checkpoint blockade. Repurposing the “flu shot” may increase response rates to immunotherapy, and based on its current FDA approval and safety profile, may be quickly translated for clinical care.
https://www.pnas.org/content/117/2/1119
発表の内容はそれぞれまちまちですが、ひとまず現時点ではICI投与中の患者でインフルエンザワクチン接種を避けることを勧めるような内容のものはなく、通常の化学療法を受ける患者と同様にインフルエンザワクチンを接種しておいて貰うのがよさそうです。
この記事に対するコメント
このページには、まだコメントはありません。
更新日:2020-11-10 閲覧数:1135 views.